NEWS: All the latest from the team
March 10, 2025
Scientific Collaboration with ARCO Technologies in Bologna, Italy
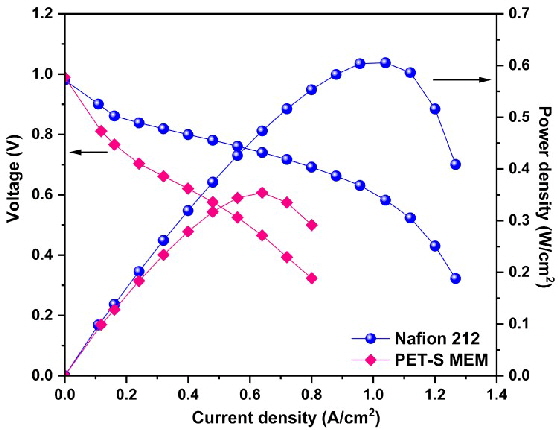
Our PhD student, Varun D N, has developed a novel proton exchange membrane (PEM) using waste PET bottles at the University of Trento. As part of the agreement with the PON project, he conducted performance tests at ARCO Technologies in Bologna, Italy. During this time, he explored catalyst preparation and gained expertise in the spray coating technique for membrane electrode assembly. He evaluated the fuel cell performance of the PET-based PEM and compared it with a commercial Nafion membrane. The results indicated promising potential for the PET-based membrane as a sustainable replacement for Nafion in fuel cell applications.
Commercial Nafion 212
Experimental PET based PEM





Measure Setup
Results

February 13, 2025
Sustainable Conversion of PET Waste Bottle into Proton Exchange Membranes for Fuel Cellseries
Varun Donnakatte Neelalochana, Ines Mancini, Nicholas Loi, Giovanni Cufalo, Angelo D’Anzi, Paolo Scardi, and Narges Ataollahi
ACS Applied Energy Materials
https://doi.org/10.1021/acsaem.4c03313
Abstract
ABSTRACT: This research introduces a sustainable method to transform poly(ethylene terephthalate) (PET) waste bottles into sulfonated proton exchange membranes for fuel cell (PEMFC) applications, addressing the need for alternatives to fluorinated materials like Nafion. The material was obtained in two steps, by amidation through a nucleophilic substitution of 4,4-diamino-2,2-stilbenedisulfonic acid (DSDA) on bis(2-hydroxyethyl) terephthalate (BHET) derived by treating PET with ethylene glycol. The membrane, prepared from the obtained product using the solution casting technique, was tested in a single fuel cell, achieving a power density of 354 mW/cm2, and compared with commercial Nafion 212 membrane (605.4 mW/cm2). The result demonstrates the potential of membrane delivered from PET as a cost-effective and sustainable solution for PEMFCs, addressing critical environmental challenges. This innovative approach transforms waste PET bottles into advanced technologies, providing a key step forward in circular economy solutions.s.

February 9, 2025
Flower-like Cu2ZnSnS4 (CZTS) transition metal sulphide (TMS) as a micro-structured electrode in rechargeable lithium batteries
M. Hassan, M.A.A.M. Abdah, P. Chelvanathan, Y. Sulaiman, F.S. Omar, H. Ahmoum, P. Scardi, M.S. Su’ait
Electrochimica Acta
https://doi.org/10.1016/j.electacta.2025.145838
Abstract
Rechargeable lithium batteries exhibit the advantages of high energy density with a broad range of potential applications, from portable electronics to highly energy-demanding electric vehicles. Recently, transition metal sulphides (TMS) have been regarded as a promising candidate for a microstructure electrode in electrochemical energy storage devices because it has a higher theoretical capacity (~847 mAh g������ 1) compared to a commercial lithium-ion battery (LIB) (~372 mAh g������ 1). Thus, this work aims to explore the capacity performance and cycling stabilities of the transition metal sulphide, Cu2ZnSnS4 (CZTS) microstructure electrode using galvanostatic technique. The CZTS was synthesised via the sol-gel method followed by sulphurisation at 550 ◦C under nitrogen atmosphere. The structural analysis showed that the CZTS nanoflower took on morphology of kesterite phase and possessed an average chemical stoichiometric composition of Cu2.2Zn1.9Sn1.5S4. The as-prepared CZTS-Li halfcell configuration exhibited high reversible capacity but lack in cycling performance at room temperature under a potential window from 3.0 to 0.01 V (vs. Li+/Li). The achieved initial discharge capacity is obtained a ~578.89 mAh g������ 1, which suggests that the kesterite CZTS can be a promising electrode material for rechargeable lithium-CZTS batteries.

February 4, 2025
Real-Time Raman Spectroscopy of Photopolymerization Dynamics in Ethylene Glycol Methyl Ether Acrylate‑g‑Epoxidized Natural Rubber
KaiLing Chai, Dhia Darwisyah Binti Dzulqurnain, Mohd Sukor Su’ait, Azizan Ahmad, Narges Ataollahi,and Tian Khoon Lee
ACS Appl. Polym. Mater. 2025, 7, 1771−1783
https://doi.org/10.1021/acsapm.4c03593
Abstract
Natural rubber (NR) is a promising material for polymer electrolytes (PEs) due to its natural abundance and desirable properties, including soft and elastic texture at room temperature, adhesive qualities, and low glass transition temperature (Tg). Additionally, it is more sustainable compared to synthetic polymers. However, its low ion transport capability poses a challenge for electrochemical applications such as lithium-ion batteries. To address this limitation, ethylene glycol methyl ether acrylate (EGMEA) was grafted onto an epoxidized natural rubber (ENR- 25) matrix at varying grafting ratios, optimizing the curing time. Photopolymerization was employed due to its rapid reaction kinetics, customizable monomer selection for tailored physical properties, and energy-efficient initiation via illumination. In situ Raman spectroscopy provided insights into the kinetics, mechanisms, and curing times of the reactions. The synthesis of EGMEA-g-ENR was confirmed through infrared (IR) and 1H NMR analyses, which validated the successful grafting of EGMEA onto ENR-25. A notable decrease in the molecular weight and polydispersity index (PDI) was observed, approaching unity, indicating improved polymer homogeneity. This decrease in molecular weight is likely due to the degradation of ENR-25 during the ultraviolet (UV)-curing process, resulting in a lower Tg (<−43 °C) compared to that of ENR-25 (−35 °C). EGMEA itself has no Tg but exhibits a Tm of −42 °C and a Tc of −65 °C. This is favorable for enhancing the ion transport in polymer electrolytes. Additionally, a significant correlation was found between the grafting efficiency and different EGMEA grafting ratios. The grafting efficiency reached up to 61.95%, with a strong correlation between the grafting efficiency and grafting ratio (R2 = 0.89). This research highlights the potential of rubber-based modification through photopolymerization for developing framework-based solid polymer electrolytes, particularly for applications in lithium-ion batteries.s.

February 1, 2025
Thermal diffuse scattering analysis of Ag2O binary system via X-ray powder diffraction
Marcelo Augusto Malagutti, Binayak Mukherjee, Himanshu Nautiyal, Sebastian Bette, Narges Ataollahi, Robert Dinnebierb and Paolo Scardi
JOURNAL OF APPLIED CRYSTALLOGRAPHY
https://doi.org/10.1107/S1600576724010756
Abstract
Diffuse scattering is a component of the powder pattern bearing information on the local atomic structure and disorder of crystalline materials. It is visible in the X-ray diffraction patterns of binary structures like Ag2O, which has a large mean squared displacement for its constituent elements. Pair distribution function (PDF) analysis is widely employed to extract this local structural information, embedded in the widths of PDF peaks. However, obtaining the PDF from experimental data requires a Fourier transform, which introduces aberrations in the transformed data due to instrument resolution, complicating the distinction between its static and dynamic components. In this work, the analysis of thermal diffuse scattering is performed directly on the X-ray powder pattern, using the traditional Rietveld method integrated with a correlated displacement model for atomic pairs. The Ag2O case study data were collected using synchrotron radiation at room temperature, supplemented by laboratory experiments up to 200°C. An Einstein model was used to obtain the harmonic and anharmonic force constants of the system. The force constants were also obtained via density functional theory and ab initio molecular dynamics simulations and showed similar values to the experiments. The analysis reveals the complex dynamic structure of Ag2O, characterized by high anisotropy in phonon dispersion relations and the presence of soft phonon modes, which explain the significant displacement parameters observed. The proposed approach can be easily employed for other binary or more complex systems to understand the dynamics of local forces through X-ray diffraction analysis.

January 20, 2025
Recycled Battery Carbon Composite Sensor for the Electrochemical Analysis of the Neurotransmitter Dopamineion
K. P. Moulya · J. G. Manjunatha · Samar A. Aldossari · Saikh Mohammad · Narges Ataollahi
Topics in Catalysis
https://doi.org/10.1007/s11244-025-02048-5
Abstract
The current article reveals a new, simple, non-toxic, sensitive, and environmentally friendly electrochemical approach for the analysis of the neurotransmitter dopamine (DP) by fabricating tartrazine (TZ) on graphite and carbon nanotube composite electrode (GP-CNTCPE) using cyclic voltammetry (CV). The graphite powder used for the analysis was extracted from a dry cell. The sensitive and selective TZ-modified composite electrode (TZ/GP-CNTCPE) displayed an effective electrochemical behavior of DP. The characteristics of both unmodified and modified sensors were evaluated using scanning electron microscopy (SEM) and electrochemical impedance studies (EIS). DP exhibited a good oxidation and reduction current response in phosphate buffered saline (PBS) of 6.5 pH. The concentration variation analysis of DP in the linear range of 0.2 μM–10.0 μM exhibited a limit of detection (LOD) of 0.39 μM and a limit of quantification (LOQ) of 1.33 μM. The anti-interference property was observed for TZ/GPCNTCPE in the existence of organic compounds and metal ions. The simultaneous analysis of DP in the presence of tyrosine (TY) indicated the good selectivity of the developed sensor. The pharmaceutical sample analysis for the presence of DP showed a fine recovery rate, and hence the developed sensor can be applicable to the real sample.

January 07, 2025
Journal Covers

This illustration depicts a thermoelectric device designed to convert thermal energy gradients into electricity. Highlighted is the nanocomposition nature of the material, that employs CuFeS2, FeS2, and Cu2S sulfide-based materials. The images were enhanced using AI assistance from ChatGPT
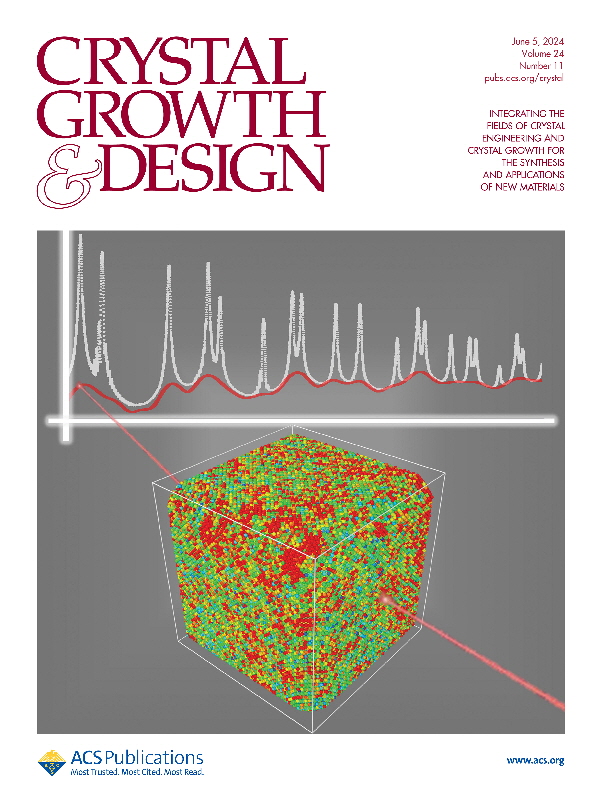
Representation of the X-ray diffraction experiment for a truncated nanocube. The colors of the atom represent the zones with high (red) and low (blue) atomic vibration correlation in the nanocrystal. The white graph is the total scattering pattern, and the red line corresponds to the thermal diffuse scattering contribution to the pattern, where the information of atomic correlation is present.